Immediate Effects of Gait Training with Rhythmic Auditory Stimulation Combined with Functional Electrical Stimulation on Gait Parameters in Patients with Subacute Stroke: A Pilot Study
© 2025 by the Korean Physical Therapy Science
Abstract
This study aimed to examine the immediate effects of rhythmic auditory stimulation and functional electrical stimulation, applied individually and in combination, on spatiotemporal gait parameters in patients with subacute stroke.
A single group repeated measures design.
Five patients diagnosed with subacute stroke participated in this study. Each participant performed three walking conditions in randomized order: walking with functional electrical stimulation, walking with rhythmic auditory stimulation, and walking with both interventions combined. Gait parameters, including gait velocity, ambulation time, number of steps, difference in step length between limbs, and gait cycle time difference, were measured using an electronic walkway analysis system. Repeated measures analysis of variance and the Friedman test were used to analyze the data.
Statistically significant differences were observed in gait velocity (F=4.487, p=.030) and ambulation time (χ²=8.143, p=.040), with the combined intervention condition showing the greatest improvement in both variables. Although post-hoc analysis did not reveal statistically significant differences, the average increase in gait velocity of 10.2 centimeters per second in the combined intervention condition approached the minimal detectable change threshold for individuals with moderate walking ability. Other variables, such as number of steps, step length differential, and cycle time differential, did not show statistically significant changes.
The findings of this pilot study suggest that the combined application of rhythmic auditory stimulation and functional electrical stimulation may lead to immediate improvements in gait performance in patients with subacute stroke. This combined approach appears to be effective in enhancing gait velocity and efficiency.
Keywords:
Functional electrical stimulation, Gait, Rhythmic auditory stimulation, Subacute strokeⅠ. Introduction
Stroke is a neurological condition that results from the interruption of cerebral blood flow or the rupture of a cerebral vessel, leading to brain damage(Shamshiev et al., 2024). As of 2016, more than 80 million people worldwide had experienced a stroke, and in South Korea, over 630,000 patients were reported as of 2022 (Health Insurance Review and Assessment Service, 2023; Shamshiev et al., 2024). Approximately half of all stroke survivors experience serious neurological deficits, one of the most common being gait disturbances caused by hemiparesis (Kelly-Hayes et al., 2003). Patients with stroke may demonstrate reduced gait speed, spatial and temporal asymmetry, and abnormal gait patterns (Hsu et al., 2003). Previous studies have identified key contributors to post-stroke gait impairment, including reduced ankle joint strength (Olney et al., 1991; Jonkers et al., 2009; Takahashi et al., 2015) and spasticity (Kitatani et al., 2016). These factors negatively affect gait symmetry, leading to inefficient and abnormal walking patterns. Many stroke patients aim to improve their gait speed and pattern as primary rehabilitation goals (Hsu et al., 2003). Although traditional lower limb strengthening programs, including the Bobath approach, have been widely applied, their effectiveness remains inconsistent.
To address slow and asymmetric gait patterns in stroke patients, RAS(rhythmic auditory stimulation; RAS) and FES(functional electrical stimulation; FES) have been proposed as effective treatment approaches (Hayden et al., 2009; Song & Ryu, 2016; Lee et al., 2018; Shan et al., 2013; Dujović et al., 2017; Bae et al., 2019). RAS, a form of neurologic music therapy, stimulates the auditory system to provide temporal structure and predictability, which can promote motor control and is commonly used for patients with gait disorders (Yoon & Kang, 2015). In contrast, FES is used to enhance neuroplasticity by facilitating repetitive, active movement training and promoting motor relearning (Kim & Kim, 2018).
Several studies have reported immediate effects of RAS and FES. For instance, Kobinata et al.(2016) found that RAS improved gait speed and step length in stroke patients, and Cha et al. (2014)observed immediate changes in speed, rhythm, step length, and symmetry depending on tempo. Additionally, Park and Wang(2017) showed that a 30-minute session of FES improved gait speed and rhythm, while Oh and Yoo(2021) reported that ankle-applied FES significantly improved gait and balance.
RAS and FES have been shown to positively affect gait speed, cadence, step length, and gait symmetry. When applied together, these two interventions may produce a synergistic effect by complementing each other’s contributions to gait improvement. However, studies that have applied both RAS and FES simultaneously are extremely limited, and no previous research has investigated their combined immediate effects.
To provide foundational data for designing future large-scale randomized controlled trials, this study was conducted as a pilot study aimed at examining the short-term intervention effects and the feasibility of the evaluation system within a limited timeframe. Accordingly, this study aimed to analyze the immediate effects of independent and combined applications of rhythmic auditory stimulation and functional electrical stimulation on gait performance in patients with stroke.
Ⅱ. Methods
1. Participants
This study included patients with subacute stroke who were hospitalized at C Hospital and M Hospital in City C. The inclusion criteria were as follows: individuals who could understand the purpose of the study and follow instructions; those who scored 24 or higher on the Korean version of the Mini-Mental State Examination (MMSE-K); and those capable of walking independently for at least 14 meters, regardless of whether they used a walking aid.
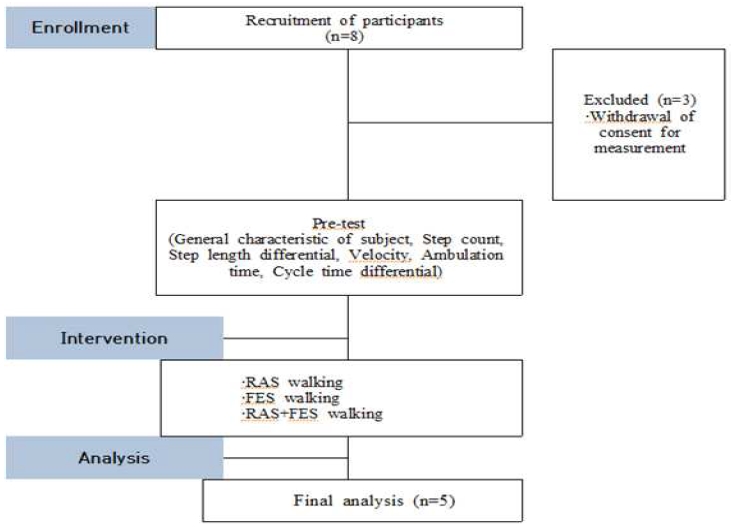
Exclusion criteria included individuals with visual or auditory impairments, cardiovascular disease, renal disease, hepatic disease, an MMSE-K score of 24 or below, cerebellar disorders, any musculoskeletal injury or surgical history that could affect gait, inability to ambulate, or refusal to participate in the study. Furthermore, individuals who exhibited physical or psychological instability during the study period, or developed orthopedic problems during the intervention, were also excluded from the final analysis. The general characteristics of the participants are presented in (Table 1.)
2. Study design
This study employed a single group repeated measures design and was conducted to analyze the immediate effects of a gait training program applying FES combined with RAS to improve spatiotemporal gait parameters in patients with stroke. The overall design of the study is illustrated in (Fig. 1).
3. Instruments
(1) FES
In this study, FES was administered using the Goscare SERA device (SERA, GOS Co., Gyeongsan, Korea, 2020)(Fig. 2.). This device is approved by the Korean Ministry of Food and Drug Safety and is classified as a Class II medical device under the name “surface electrode-type functional muscle electrical stimulation equipment.”
The SERA device is compact and discreet in appearance, making it aesthetically favorable when worn. Weighing approximately 457 grams, it is lightweight and comfortable to wear even during extended activity. This device provides electrical stimulation to induce muscle contraction during the swing phase based on input from a foot sensor inserted into the shoe, thereby preventing foot drop.
(2) RAS
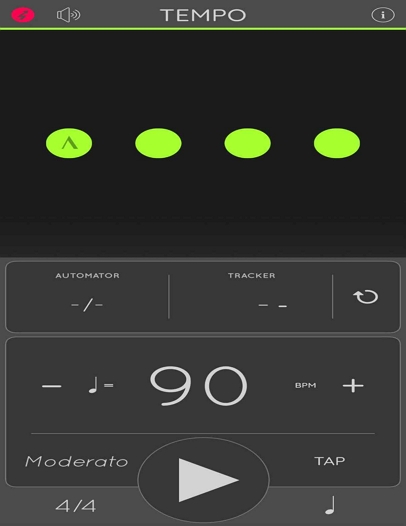
In this study, RAS was provided using a smartphone metronome application (Metronome: Tempo Lite, Frozen Ape, Singapore, 2009), as shown in (Fig. 3). The metronome is a device that emits regular auditory beats. In this study, the rhythmic cues were applied at the initiation of the swing phase of the paretic lower limb (Lee et al., 2011). The beats per minute (BPM) were individually adjusted based on each participant’s self-selected gait speed (Ghai et al., 2018).
(1) GaitRite
In this study, the GAITRite system(GAITRite, CIR Systems Inc, Franklin NJ, USA, 2008), a validated and reliable tool, was used to objectively assess spatiotemporal gait variables and plantar pressure distribution. The GAITRite system consists of an electronic walkway with a total length of 488 cm, a sensing area of 366 cm in length and 61 cm in width. The mat contains 18,432 pressure sensors arranged in a grid of 48 columns and 384 rows, with a sensor spacing of 1.27 cm.
When the participant walks on the mat, the sensors detect foot pressure and transmit the data to a computer via a serial interface cable. This system is portable and can be installed on any flat surface. It offers the advantage of providing accurate and objective data from a single walking trial, thereby enhancing participant convenience.
The reliability of the GAITRite system has been reported to be excellent, with an intraclass correlation coefficient (ICC) of 0.95 for gait velocity and 0.96 for cadence (McDonough et al., 2001). In this study, participants walked a total of 14 meters, and measurements were taken beginning from the 5-meter mark.
4. Experimental Procedure
Participants were recruited and selected according to inclusion and exclusion criteria. Prior to the intervention, each participant's basic demographic information, step count, step length differential, velocity, ambulation time, and cycle time differential were measured using the GaitRite system. During this assessment, participants were asked to walk 14 meters at their usual walking speed to measure their self-selected gait velocity.
The intervention consisted of three conditions: FES walking, RAS walking, and combined RAS+FES walking. To minimize learning effects and potential bias related to the order of conditions, the sequence of the three conditions was randomized for each participant, and all interventions were conducted individually in an independent, isolated space.
In the FES condition, FES was applied to the ankle dorsiflexor muscles during walking. The electrodes were attached to the tibialis anterior (anode) on the paretic side and over the common peroneal nerve near the fibular head (cathode), generating functional movement that facilitated appropriate ankle dorsiflexion while minimizing excessive inversion. The stimulation parameters were set as a rectangular waveform with a pulse width of 150 μs and a frequency of 25 Hz. The intensity was adjusted to the maximum level tolerable by each participant, depending on their level of functional recovery (Lindquist, Prado et al., 2007). In the RAS condition, a metronome beat was provided based on the cadence of the self-selected gait velocity collected using the GaitRite system during the pre-test. The metronome beat was delivered in a 4/4 rhythm, synchronized with the initiation of the swing phase of the paretic lower limb, and participants were instructed to walk in time with the swing phase of their paretic limb. In the combined RAS+FES condition, both RAS and FES were applied simultaneously (Fig. 4.). Participants were similarly instructed to synchronize their gait with the metronome beat during the swing phase of the paretic limb, while the FES automatically detected and synchronized with the swing phase of the paretic limb.
Adequate rest periods were given between conditions to prevent participant fatigue. After completing data collection for all intervention conditions, spatiotemporal gait changes before and after the interventions were analyzed. This allowed for the assessment of the immediate effects of RAS and FES on participants’ gait characteristics. The collected data were statistically analyzed to compare differences between the intervention conditions and to systematically evaluate their effects.
5. Data Analysis
All data analyses were performed using SPSS Statistics version 27.0. Descriptive statistics were used to summarize the general characteristics of the participants. To assess the normality of the dependent variables, the residuals of each variable were tested using the Shapiro–Wilk test.
For variables that satisfied the assumption of normality, repeated measures analysis of variance (ANOVA) was used to evaluate intervention effects. For those that did not meet the normality assumption, the Friedman test was employed.
Post-hoc comparisons between intervention conditions were conducted using the Bonferroni correction method. The statistical significance level was set at α = 0.05 for all analyses
Ⅲ. Results
1. Changes in Spatiotemporal Gait Parameters
Repeated measures ANOVA revealed a statistically significant difference in gait velocity (cm/sec) across the conditions (F = 4.487, p = .03). indicating a tendency toward increased gait speed(Table 2). However, post-hoc analysis using the Bonferroni correction revealed no statistically significant differences between conditions (p > .05), although the combined FES and RAS condition showed the highest gait velocity, it did not reach statistical significance (Table 3). No significant differences were found in step count (p = .12) or step length differential (p = .34) (Table 2).
For variables that did not meet the assumption of normality, the Friedman test was conducted. A significant difference was found in Ambulation Time (sec) (χ² = 8.143, df = 3, p = .04). The mean ranks were 3.20 in the pre-intervention condition, 3.30 in the FES condition, 2.20 in the RAS condition, and 1.30 in the combined condition, indicating the shortest ambulation time in the combined condition (Table 4). No significant difference was found in Cycle Time Differential (p = .22) (Table 4). Post-hoc comparisons with Bonferroni correction showed a trend toward decreased ambulation time in the combined condition compared to the pre-intervention condition, although this difference was not statistically significant (p = .120). A similar decreasing trend was also observed between the FES and combined conditions, but again, it was not statistically significant (p = .086) (Table 5).
Ⅳ. Discussion
This preliminary study aimed to examine the immediate effects of RAS and FES, applied individually and in combination, on gait parameters in patients with stroke. A total of five individuals with subacute stroke participated in the study.
Based on the study results, statistically significant differences in gait velocity and ambulation time were found across all intervention conditions. However, post-hoc tests did not reveal statistically significant differences between individual conditions. Furthermore, the lack of statistically significant differences between individual conditions in the Bonferroni post-hoc test can be attributed to several factors.First, the very small sample size in this study likely resulted in limited statistical power, making it difficult to detect existing differences between interventions. Second, since all interventions—FES, RAS, and the combined FES+RAS—positively influenced gait performance, the relative differences between conditions may have been modest. Additionally, the Bonferroni correction applies a highly conservative adjustment to control for Type I error arising from multiple comparisons, thereby increasing the risk of Type II error, particularly in studies with small sample sizes or small effect sizes. Considering these factors, although statistical significance was not clearly established, the observed changes may still be interpreted as clinically meaningful.
Gait velocity increased compared to baseline under all intervention conditions FES, RAS, and the combined FES+RAS with the greatest improvement observed under the combined condition. Ambulation time decreased compared to baseline in the RAS and combined FES+RAS conditions, with the most pronounced reduction observed under the combined condition. This finding may be attributed to a complementary interaction between the two interventions.
RAS facilitates the preparation for subsequent movements and promotes sequential muscle activation, thereby enhancing the rhythmicity and regularity of gait (Thaut et al., 1997). FES, when applied to the tibialis anterior of the paretic side, corrects foot drop and supports the swing phase of gait (Lindquist et al., 2007). Lindquist et al. (2007) reported that FES applied to the paretic tibialis anterior muscle promotes coordinated lower limb movements in stroke patients.
Importantly, FES goes beyond mere mechanical assistance by delivering repetitive, functional sensory-motor input synchronized with the gait cycle, which may stimulate neuroplasticity. This patterned stimulation facilitates coordinated activation of lower limb muscles and promotes the reorganization of the motor cortex and subcortical regions, thereby accelerating functional recovery (Zheng et al., 2018). These trends are consistent with previous studies by Kobinata et al. (2016), Park and Wang (2017), Oh and Yoo (2021), and Kim and Kim (2018). This study is distinguished from previous research in that it directly compared the immediate effects of combined application of RAS and FES, rather than applying them individually, based on the existing theoretical background. While most prior studies focused on the effects of either RAS or FES alone, this study aimed to investigate the synergistic effects on gait function when both interventions were applied simultaneously. As a result, although statistical significance was not achieved under the RAS+FES combined condition, the greatest increase in gait speed and the most substantial reduction in gait time compared to baseline were successfully observed. Furthermore, unlike the study by Kim and Kim (2018), which examined the long-term effects of a four-week gait training program combining RAS and FES, the present study focused on comparing the immediate effects following intervention. These findings suggest that the combined application of RAS and FES may yield not only long-term benefits through extended training but also immediate effects in clinical settings.
Although the Bonferroni post-hoc test did not reveal statistically significant differences, the approximately 10 cm/s increase in gait velocity under the FES+RAS condition, compared to the pre-intervention baseline, may be considered clinically meaningful. According to a study by Hosoi et al. (2023), the MDC(minimum detectable change) in stroke patients varies depending on their baseline gait speed. Therefore, the interpretation of treatment effects should be individualized based on each participant’s gait velocity.
In the present study, the participants’ mean pre-intervention gait speed was 0.74 m/s, indicating a moderate gait capacity. The MDC for individuals with moderate gait speed is reported to be 0.11 m/s. Given that the average velocity under the FES+RAS condition reached 0.84 m/s, the observed improvement approached the MDC threshold, supporting its clinical relevance(Hosoi et al., 2023).
Gait speed is a well-established predictor of functional recovery and return to independent living in stroke patients (Khanittanuphong & Tipchatyotin, 2017; Yamada et al., 2018; Grau-Pellicer et al., 2019; Zhao et al., 2024). Thus, the observed change may be interpreted as an early indicator of functional improvement.
In addition, ambulation time—a variable closely related to gait velocity—also showed a meaningful decrease, with the shortest time recorded under the FES+RAS condition. This suggests that the combined intervention may enhance gait efficiency more effectively than single interventions, indicating potential improvements in real-world mobility for stroke patients. However, statistical significance was not achieved in post-hoc comparisons, likely due to the small sample size and inter-individual variability.
In contrast, no statistically significant differences were found in step count, step length differential, or cycle time differential, which are variables related to the precision and symmetry of gait. These variables are primarily associated with fine motor control mechanisms of walking that require bilateral coordination and balanced propulsion. Improvements in such aspects are more likely to result from repeated and sustained interventions rather than a single-session stimulation.
In particular, step length differential is a key indicator of gait symmetry. A study by Schwartz et al. (2022) identified the Step Length SI(Symmetry Index) as a significant predictor of rehabilitation duration in patients with stroke. In that study, SI was found to be significantly correlated with various functional indicators such as the 10MWT(10-Meter Walk Test), NIHSS(National Institutes of Health Stroke Scale), FIM(Functional Independence Measure), and FAC(Functional Ambulation Category), and was reported to be an even stronger predictor of rehabilitation duration than gait velocity. These findings suggest that gait symmetry is influenced not only by temporal or velocity improvements, but also by broader aspects of functional recovery and long-term rehabilitation. The limited immediate change in step length differential observed in this study may reflect the nature of the variable, which is more responsive to long-term neuromotor adaptation than to short-term stimulation. Overall, the findings of this preliminary study suggest that combined RAS and FES interventions have potential for immediate improvements in gait performance.
This study has several limitations. First, the small sample size (N = 5) limited statistical power, and results should be interpreted with caution when generalizing. Second, the study only investigated immediate effects; therefore, medium- or long-term changes resulting from continuous interventions were not addressed. Third, the RAS applied in this study used a fixed tempo based on each participant’s self-selected gait speed, which may have limited the full potential of RAS to elicit responses through tempo variations.
Nevertheless, this study suggests that the combined intervention of RAS and FES may have immediate clinical efficacy in improving gait performance in patients with subacute stroke. It also highlights the importance of prioritizing function-oriented indicators, such as gait speed and ambulation time, when designing intervention strategies. As a pilot study, the present findings help identify potential methodological limitations and areas requiring refinement, thereby providing an initial basis for evaluating the feasibility and validity of future large-scale research.
Future studies should involve larger sample sizes, various RAS tempo conditions, and repeated intervention designs. Such expanded research may ultimately contribute to the development of effective and practical gait rehabilitation programs in clinical settings.
Ⅴ. Conclusion
This preliminary study aimed to investigate the immediate effects of RAS and FES, applied individually and in combination, on gait parameters in patients with stroke. The findings demonstrated that the combined FES+RAS intervention produced significant immediate effects, including increased gait velocity and reduced ambulation time, suggesting its potential as a short-term intervention strategy for improving gait performance in subacute stroke patients.
Although no significant changes were observed in variables related to gait symmetry—such as step count, step length differential, and cycle time differential—these aspects may require gradual improvement through repeated training over time.
References
-
Bae DY, Shin JH, Kim JS. Effects of dorsiflexor functional electrical stimulation compared to an ankle/foot orthosis on stroke-related genu recurvatum gait. J Phys Ther Sci. 2019;31:865-868.
[https://doi.org/10.1589/jpts.31.865]

-
Cha Y, Kim Y, Chung Y. Immediate effects of rhythmic auditory stimulation with tempo changes on gait in stroke patients. J Phys Ther Sci. 2014;26:479-482.
[https://doi.org/10.1589/jpts.26.479]

-
Dujović SD, Malešević J, Malešević N, et al. Novel multi-pad functional electrical stimulation in stroke patients: a single-blind randomized study. NeuroRehabilitation. 2017;41(4):791-800.
[https://doi.org/10.3233/NRE-172153]

-
Ghai S, Ghai I, Effenberg AO. Effect of rhythmic auditory cueing on aging gait: a systematic review and meta-analysis. Aging Dis. 2018;9(5):901.
[https://doi.org/10.14336/AD.2017.1031]

-
Grau-Pellicer M, Chamarro-Lusar A, Medina-Casanovas J, et al. Walking speed as a predictor of community mobility and quality of life after stroke. Top Stroke Rehabil. 2019;26:349-358.
[https://doi.org/10.1080/10749357.2019.1605751]

-
Hayden R, Clair A, Johnson G, et al. The effect of rhythmic auditory stimulation (RAS) on physical therapy outcomes for patients in gait training following stroke: a feasibility study. Int J Neurosci. 2009;119:2183-2195.
[https://doi.org/10.3109/00207450903152609]

- Health Insurance Review and Assessment Service (HIRA). Current status of treatment for cerebrovascular diseases. 2023.
-
Hosoi Y, Kamimoto T, Sakai K, et al. Estimation of minimal detectable change in the 10-meter walking test for patients with stroke: a study stratified by gait speed. Front Neurol. 2023;14:1219505.
[https://doi.org/10.3389/fneur.2023.1219505]

-
Hsu A L, Tang P F, Jan M H. Analysis of impairments influencing gait velocity and asymmetry of hemiplegic patients after mild to moderate stroke. Arch Phys Med Rehabil. 2003;84(8):1185-1193.
[https://doi.org/10.1016/S0003-9993(03)00030-3]

-
Jonkers I, Delp S, Patten C. Capacity to increase walking speed is limited by impaired hip and ankle power generation in lower functioning persons post-stroke. Gait Posture. 2009;29(1):129-137.
[https://doi.org/10.1016/j.gaitpost.2008.07.010]

-
Kelly-Hayes M, Beiser A, Kase C S, et al. The influence of gender and age on disability following ischemic stroke: the Framingham study. J Stroke Cerebrovasc Dis. 2003;12(3):119-126.
[https://doi.org/10.1016/S1052-3057(03)00042-9]

-
Khanittanuphong P, Tipchatyotin S. Correlation of the gait speed with the quality of life and the quality of life classified according to speed-based community ambulation in Thai stroke survivors. NeuroRehabilitation. 2017;41(1):135-141.
[https://doi.org/10.3233/NRE-171465]

-
Kim SH, Kim YM. Effects of Functional Electrical Stimulation Gait Training with Rhythmic Auditory Stimulation on Balance and Gait Ability of Stroke Patients. J Korean Soc Phys Med. 2018;13(4):95-103.
[https://doi.org/10.13066/kspm.2018.13.4.95]

-
Kitatani R, Ohata K, Sato S. et al. Ankle muscle coactivation and its relationship with ankle joint kinematics and kinetics during gait in hemiplegic patients after stroke. Somatosens Mot Res. 2016;33(2):79-85.
[https://doi.org/10.1080/08990220.2016.1178636]

-
Kobinata N, Ueno M, Imanishi Y, et al. Immediate effects of rhythmic auditory stimulation on gait in stroke patients in relation to the lesion site. J Phys Ther Sci. 2016;28:2441-2444.
[https://doi.org/10.1589/jpts.28.2441]

-
Lee SH, Lee K, Song C. Gait Training with Bilateral Rhythmic Auditory Stimulation in Stroke Patients: A Randomized Controlled Trial. Brain Sci. 2018;8.
[https://doi.org/10.3390/brainsci8090164]

-
Lee SH, Lee KJ, Ha GH. et al. The effects of rhythmic auditory stimulation on the gait symmetry in the chronic stroke patients. J Korea Acad Ind Coop Soc. 2011;12(5):2187-2196.
[https://doi.org/10.5762/KAIS.2011.12.5.2187]

-
Lindquist A R, Prado C L, Barros R M, et al. Gait training combining partial body-weight support, a treadmill, and functional electrical stimulation: effects on poststroke gait. Phys Ther. 2007;87(9):1144-1154.
[https://doi.org/10.2522/ptj.20050384]

-
McDonough A L, Batavia M, Chen F C. et al. The validity and reliability of the GAITRite system's measurements: A preliminary evaluation. Arch Phys Med Rehabil. 2001;82(3):419-425.
[https://doi.org/10.1053/apmr.2001.19778]

-
Oh DG, Yoo K. Effects of Functional Electrical Stimulation (FES) on the Temporal-spatial Gait Parameters and Activities of Daily Living in Hemiplegic Stroke Patients. J Korean Soc Phys Med. 2021;16(3):37-44.
[https://doi.org/10.13066/kspm.2021.16.3.37]

- Olney S J, Griffin M P, Monga T N. et al. Work and power in gait of stroke patients. Arch Phys Med Rehabil. 1991;72(5):309-314.
-
Park SJ, Wang J. The immediate effect of FES and TENS on gait parameters in patients after stroke. J Phys Ther Sci. 2017;29:2212-2214.
[https://doi.org/10.1589/jpts.29.2212]

-
Schwartz I, Ofran Y, Karniel N. et al. Step Length Asymmetry Predicts Rehabilitation Length in Subacute Post Stroke Patients. Symmetry. 2022;14:1995.
[https://doi.org/10.3390/sym14101995]

- Shamshiev A, Saduakas A, Zhakubaev M A. et al. STROKE: A COMPREHENSIVE OVERVIEW OF TRENDS, PREVENTION, AND TREATMENT (LITERATURE REVIEW). Bull Surg Kazakhstan. 2024
- Shan S, Huang G, Zeng Q.Effects of gait triggered functional electrical stimulation on temporal-spatial parameters of gait in foot drop patients after stroke. Chinese Journal of Rehabilitation Medicine. 2013;28:558–563.
-
Song GB, Ryu H. Effects of gait training with rhythmic auditory stimulation on gait ability in stroke patients. J Phys Ther Sci. 2016;28:1403-1406.
[https://doi.org/10.1589/jpts.28.1403]

-
Takahashi K Z, Lewek M D. Sawicki G S. A neuromechanics-based powered ankle exoskeleton to assist walking post-stroke: a feasibility study. J Neuroeng Rehabil. 2015;12:1-13.
[https://doi.org/10.1186/s12984-015-0015-7]

-
Thaut M H, McIntosh G C, Rice R R. Rhythmic facilitation of gait training in hemiparetic stroke rehabilitation. J Neurol Sci. 1997;151(2):207-212.
[https://doi.org/10.1016/S0022-510X(97)00146-9]

-
Yamada R, Ichinosawa Y, Shimizu S. et al. Gait speed is a strong predictor of the ability to walk independently without wheelchair assistance in a facility setting for post-stroke patients. Ann Phys Rehabil Med. 2018;61:e218
[https://doi.org/10.1016/j.rehab.2018.05.505]

-
Yoon S. Kang S. Effects of inclined treadmill walking training with rhythmic auditory stimulation on balance and gait in stroke patients: a pilot study. J Korean Soc Integr Med. 2015;3(4):69-78.
[https://doi.org/10.15268/ksim.2015.3.4.069]

-
Zhao Y, Liao X, Gu H. et al. GAIT SPEED AT THE ACUTE PHASE PREDICTED HEALTH-RELATED QUALITY OF LIFE AT 3 AND 12 MONTHS AFTER STROKE: A PROSPECTIVE COHORT STUDY. J Rehabil Med. 2024;56:24102
[https://doi.org/10.2340/jrm.v56.24102]

-
Zheng X Y, Chen D, Yan T. et al. A Randomized Clinical Trial of a Functional Electrical Stimulation Mimic to Gait Promotes Motor Recovery and Brain Remodeling in Acute Stroke. Behav Neurol. 2018;(1):8923520
[https://doi.org/10.1155/2018/8923520]